Virus Packaging
Virus Packaging
Fast, high-titer virus ready to use
We offer express lentiviral and retroviral packaging services to produce high-quality, high-titer virus particles using your viral construct with a turnaround time of 1-2 weeks.
Highlighted Features
Time Period
1-2 Weeks (10 business days)
Deliverables
5 x 20 μL (100 μL) or 10 x 20μL (200 uL)
Titer
Regular: 107 IFU/mL
High: 108 IFU/mL
Ultra High: 109 IFU/mL
Quality Control
qPCR to measure infectious units per ml (IFU/ml)
Optional: Plasmid amplification, custom vector building, and large-scale production. Please CONTACT US for any special requests.
Background
Lentiviral and retroviral vectors are one of the most efficient vehicles for gene transfer in mammalian cells both in vitro and in vivo. Lentiviral vectors are capable of infecting non-dividing and dividing cells, whereas retroviral vectors can only infect dividing cells. This means that lentivirus can infect a greater variety of cell types than retrovirus.
To increase the safety of using lentivirus, the components necessary for virus production are split across multiple plasmids (3 for 2nd-generation systems, 4 for 3rd-generation systems). The components of both systems are as follows:
- Lentiviral transfer plasmid encoding your insert of interest. The transgene sequence is flanked by long terminal repeat (LTR) sequences, which facilitate integration of the transgene sequence into the host genome. In general, the sequences between and including the LTRs will integrate into the host genome upon viral transduction. Most of lentiviral transfer plasmids are based on the HIV-1 virus. For safety concerns, the transfer plasmids should be replication incompetent and contain an additional deletion in the 3'LTR, rendering the virus "self-inactivating" (SIN) after transgene integration.
- Packaging plasmids contains the Gag, Pol and Rev genes.
- VSV‐G protein is standard for the envelope plasmid due to its wide infectivity.
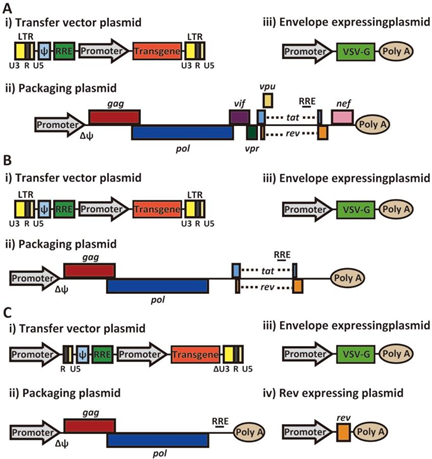
Schematic representation of the three generations of lentiviral packaging constructs: A) First generation packaging system. B) Second generation packaging system. C) Third generation packaging system.
The system of three expression cassettes developed in 1996 is considered the first generation of lentivirus. In this system the packaging cassette has all structural proteins, with exception of Env glycoproteins, and all accessory and regulatory proteins. The 5' LTR was substituted by a strong promoter (CMV or RSV) and the 3' LTR by an SV40 or insulin poly(A) site to reduce the homology between the cassettes. In the Env expression cassette the gp120 from HIV-1 was replaced by other envelope genes such as VSV-G or amphotrophic MLV. The transgene cassette was composed of the 5' LTR, the ψ with a truncated gag gene, the RRE cis-acting region and the gene of interest under the control of a heterologous promoter (usually CMV) and the 3'LTR. This system allowed an easy way to achieve good titers but its level of safety was not very high.
In the second generation lentiviral system, the three plasmid system was maintained, but all accessory genes not required for viral replication in vitro (vif, vpr, vpu, and nef) were removed, improving safety and decreasing cytotoxicity without negative effects on vector yield or infectivity.
In the third generation lentiviral system, the tat is eliminated and the rev is placed in an independent plasmid. The tar sequence was replaced by a strong heterologous promoter. Therefore Tat protein was no longer necessary to increase the transgene transcription and the tat gene was eliminated. Rev was placed in an independent non-overlapping plasmid increasing the safety as now four events of homologous recombination would be required for replication competent lentivirus (RCL) formation. With these new features, the vectors of the third-generation system offer a higher level of biosafety and. Additionally, since the resulting titers do not decrease, third generation vectors have become the most commonly used lentivirus system.
Through years of experience with lentiviral and retroviral vectors, we have developed our own proprietary virus packaging systems and efficient protocols for rapid generation of pseudoviral particles. Our lentiviral transfer plasmids, and packaging mix are generalized and appropriate for wide-ranging cell types and systems. The process of producing infectious transgenic lentivirus is outlined in the simple schema below.
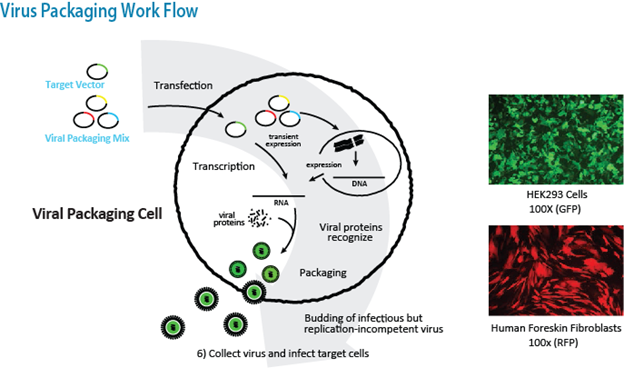
Expression and packaging mix plasmids are transfected into HEK293FT cells: one transfer plasmid, one or two packaging plasmid(s) and one envelope plasmid. After a media change and an incubation period of approximately 48 hours, supernatant containing the virus is removed and stored, or centrifuged to concentrate virus. Crude or concentrated virus can then be used to transduce the cells of interest.
IFU Determination
Once viral supernatant or concentrated virus is collected, genomic viral RNA is purified from a small aliquot of the virus using a viral RNA purification kit. The RNA is then treated with DNase I to remove any residual plasmid DNA carried over from transient transfection of the packaging cells. Serial dilutions of the viral RNA sample are subjected to qRT-PCR to determine threshold cycle (Ct) values for each dilution. The RNA genome copy number in a sample dilution is determined by finding the copy number that corresponds to its Ct value on a standard curve generated from serial dilutions of the calibrated Lenti-X RNA Control Template.
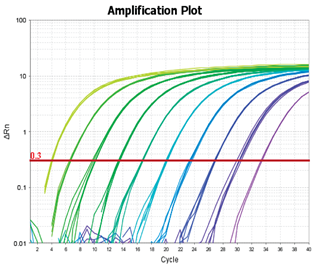
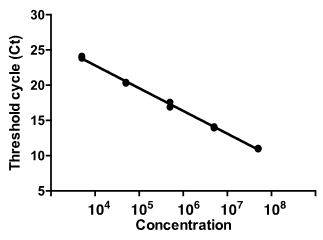
Representative RT-qPCR analysis used for titration of HIV-1-based lentivirus. Genomic RNA extracted from lentivirus vectors was subjected to qPCR amplification and monitored using a QuantStudio 3 (Applied Biosystems) with sets of primers specific for HIV Gag. Amplification plots were displayed and cycle threshold values (Ct) were measured (left panel). SDS software allows calculation of the standard curve and determination of the Gag quantity for each sample (right panel).
An IFU value is also determined by independent means (e.g. via FACS or colony selection) to establish a relationship between the two values. For subsequent, similarly prepared virus supernatants, the qRT-PCR titration value may be used as a reference to determine a relative IFU value for the concentrated virus and then later, the MOI for the infection experiment.
Publications
- Mitochondrial Fission Promotes the Continued Clearance of Apoptotic Cells by Macrophages. Wang Y et al. Cell. 2017 Oct 5;171(2):331-345.
- All-optical synaptic electrophysiology probes mechanism of ketamine-induced disinhibition, Linlin Z. Fan et al. Nat Methods. 2018 Oct; 15(10): 823–831.
- Detection of Aggregation-Competent Tau in Neuron-Derived Extracellular Vesicles, Francesc X. Guix et al. Int J Mol Sci. 2018 Mar; 19(3): 663.
Ordering Information
If you don't see the titer or volume your project requires, please CONTACT US for a price request.
| Service | Titer | Volume | IFU/mL | Catalog # | Price |
|---|---|---|---|---|---|
| Lentivirus | Regular Titer | 100 µl | >107 | CS-LV03 | Inquire |
| Lentivirus | Regular Titer | 200 µl | >107 | CS-LV03X | Inquire |
| Lentivirus | High Titer | 100 µl | >108 | CS-LV02 | Inquire |
| Lentivirus | High Titer | 200 µl | >108 | CS-LV02X | Inquire |
| Lentivirus | Ultra-High Titer | 100 µl | >109 | CS-LV01 | Inquire |
| Lentivirus | Ultra-High Titer | 200 µl | >109 | CS-LV01X | Inquire |
| Retrovirus | Regular Titer | 100 µl | >107 | CS-RV03 | Inquire |
| Retrovirus | Regular Titer | 200 µl | >107 | CS-RV03X | Inquire |
| Retrovirus | High Titer | 100 µl | >108 | CS-RV02 | Inquire |
| Retrovirus | High Titer | 200 µl | >108 | CS-RV02X | Inquire |
| Retrovirus | Ultra-High Titer | 100 µl | >109 | CS-RV01 | Inquire |
| Retrovirus | Ultra-High Titer | 200 µl | >109 | CS-RV01X | Inquire |
Have Questions?
If you don't see the product you're looking for, please don't hesitate to CONTACT US. Our dedicated team of scientists will provide you our best solutions for your desired results.

